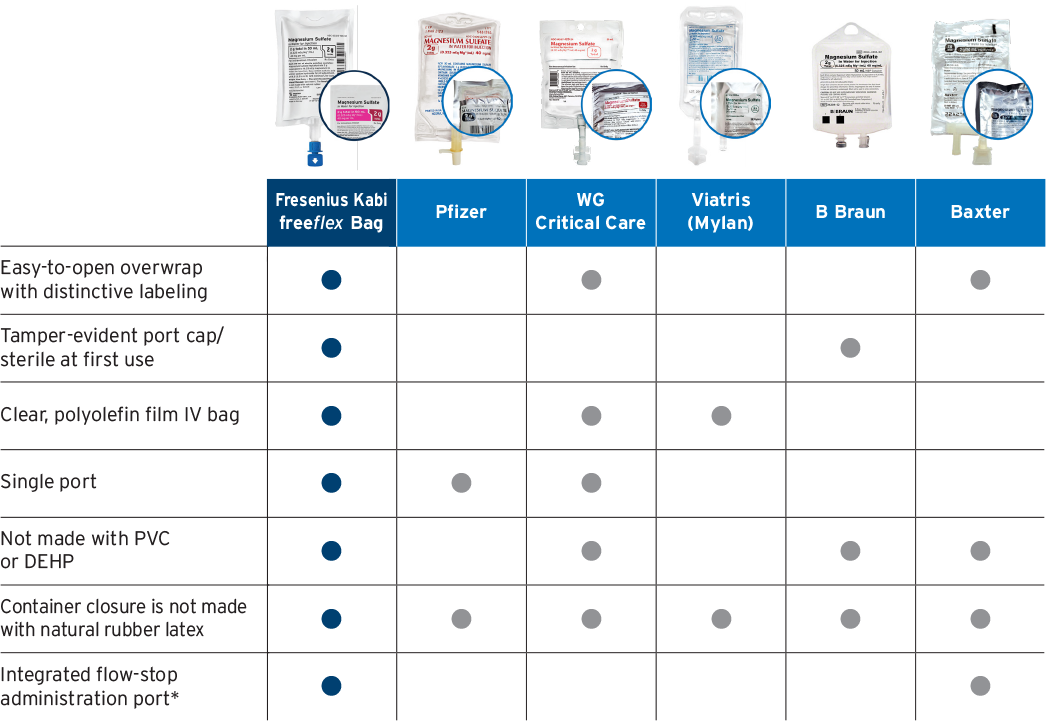
According to the Institute for Safe Medication Practices (ISMP): To the extent possible, using commercially manufactured parenteral products over manually compounded sterile preparations is a Best Practice for Sterile Compounding.1
Premix: An ISMP best practice
Key Advantages of Premix
FDA-approved safety and consistency
Premix medications are manufactured under full cGMP oversight and undergo FDA review for safety, efficacy, and quality before release. This helps support consistent doses, predictable stability, and standardized labeling.
Reduced workflow burden
By minimizing manual sterile compounding, premix can help reduce preparation time2 and free pharmacy resources for clinical services or higher-priority compounding.
Supply reliability
Premix products come with long shelf lives (18-36 months) and can be purchased through standard wholesale channels, helping provide predictable availability and less waste.
Clinician and patient safety
Fewer manual manipulations mean fewer opportunities for contamination, needlesticks, or accidental exposure.2 Premix also offers standardized strengths and concentrations that
support medication administration and help reduce potential for error.
Why Premix Matters Fact Sheet
View PDF
How Premix Complements Compounding
Compounding pharmacies play an important role, especially when treating patients who require individualized therapies or during drug shortages.
The addition of premix can support them by:
- Reducing routine or high‑volume compounding so pharmacies can prioritize specialty or patient‑specific preparations.
- Providing a consistent baseline product that helps reduce variability across shifts, staff, and sites.
- Allowing compounding teams to maintain focus on the cases where their expertise is most needed.
Manufactured vs. Compounded Medications
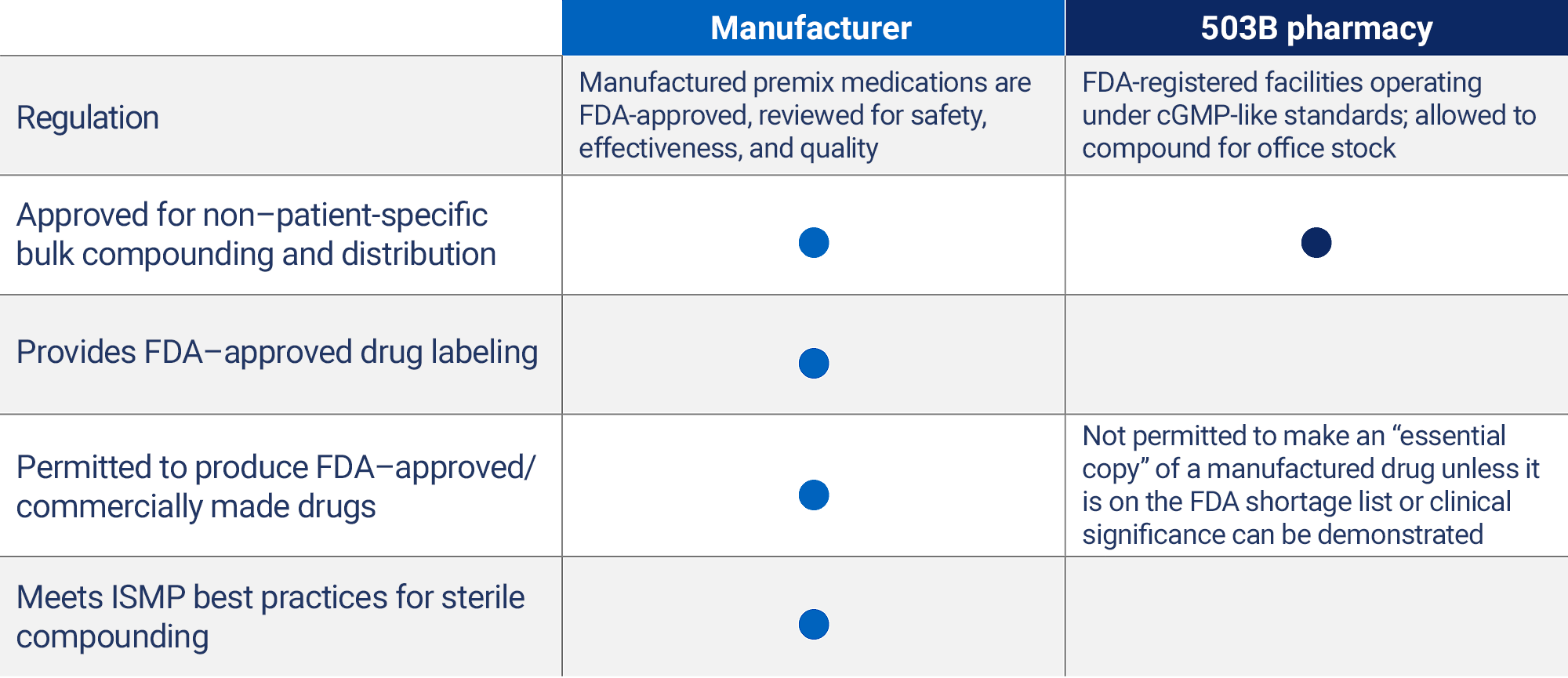
Compounding remains a critical pharmacy function. Premix enhances this capability by offering predictable, ready-to-use, FDA‑approved medications that help support operational efficiency, patient safety, and adherence to best practices.
By using premix where appropriate, pharmacies may be able to better balance safety, workload, and clinical priorities, focusing compounding expertise where it makes the greatest impact.
Compounding errors to avoid
In a 2020 survey3, the ISMP found that nearly 75% of respondents knew of at least one pharmacy sterile compounding error in the past year, such as:
- Incorrect dose or concentration (58%)
- Incorrect base solution (51%)
- Incorrect base solution volume (43%)
- Incorrect reconstitution of a drug (volume or diluent) (36%)
- Incorrect drug (35%)
- Expired drug, base solution, or compounded sterile preparation (16%)
- Omission of a drug (5%)
References
1. 2022 ISMP Guidelines for Sterile Compounding and the Safe Use of Sterile Compounding Technology, page 17.
2. AJ Loeb, DA Fishman, TR Kochis. Premixed intravenous admixtures: a critical challenge for hospital pharmacy. Am J Hosp Pharm 1983 Jun;40(6):1041-3. Accessed January 3, 2024.
3. Institute for Safe Medication Practices, October 22, 2020. ISMP Survey Provides Insights into Pharmacy Sterile Compounding Systems and Practices. Accessed at https://home.ecri.org/blogs/ismp-news/ismp-survey-on-sterile-compounding-in-pharmacies-reveals-gaps-challenges on January 3, 2024.